Ep Catalogue
Ep Catalogue - Search by catalogue code, name, monograph or cas number to find the standard you need. This document provides a list of european pharmacopoeia reference standards including the order, reference standard code, effective date, batch number, quantity per vial, sale unit,. The european directorate for the quality of medicines & healthcare provides access to reference standards, safety data sheets, and information leaflets. Access the electronic version of the european pharmacopoeia, the official compendium of pharmaceutical standards in europe. The edqm also provides batch validity statements, safety data sheets and. Search for crs (certified reference substances) by name or code in the european pharmacopoeia catalogue. Find and buy online access to technical information for vehicle industry, such as spare part catalogues, workshop manuals, diagnostic programs and service manuals. The united states pharmacopeia (usp), the european. Rs catalogue is updated on a daily basis and gives access not only to all the ph. Find out the latest news, orders, catalogues and safety data sheets. Find information on batch number, unit quantity, price, shipping conditions and. This catalogue is updated on a daily. The edqm also provides batch validity statements, safety data sheets and. Eur.) offers a portfolio of more than 2 800 reference standards. Erythromycin c is a catalogue code for a reference standard used in european pharmacopoeia monographs. Our new interactive 2025 catalogue is now available, offering instant access to sample data, scheme descriptions, application forms, and videos, plus a search function to quickly find what. Find and buy online access to technical information for vehicle industry, such as spare part catalogues, workshop manuals, diagnostic programs and service manuals. Batch validity statements (bvss) for each reference standard;. Find out how to subscribe, consult, and get help with the. We distribute chemical reference substances and reference standards from the world’s three leading pharmacopoeias: Find information on batch number, unit quantity, price, shipping conditions and. Access the electronic version of the european pharmacopoeia, the official compendium of pharmaceutical standards in europe. Learn how to participate in establishing and using the. Edqm provides reference standards for the quality control of medicines and health care products in europe and worldwide. Please enter a search term and. This document provides a list of european pharmacopoeia reference standards including the order, reference standard code, effective date, batch number, quantity per vial, sale unit,. For example, if you enter. Please enter a search term and select a search method using the drop menus below. Find and order reference standards for the european pharmacopoeia monographs from the edqm. Our new. Batch validity statements (bvss) for each reference standard;. We distribute chemical reference substances and reference standards from the world’s three leading pharmacopoeias: Access the full list here. Edqm provides reference standards for the quality control of medicines and health care products in europe and worldwide. Find information on batch number, unit quantity, price, shipping conditions and. Find information on batch number, unit quantity, price, shipping conditions and. Download the pdf catalogue or use the online database with updated information and. Edqm provides reference standards for the quality control of medicines and health care products in europe and worldwide. The european directorate for the quality of medicines & healthcare provides access to reference standards, safety data sheets,. If you select contains, all entries containing your search term will be returned. Learn how to participate in establishing and using the. Edqm provides reference standards for the quality control of medicines and health care products in europe and worldwide. The catalogue of the european pharmacopoeia (ph. The united states pharmacopeia (usp), the european. The united states pharmacopeia (usp), the european. Download the pdf catalogue or use the online database with updated information and. Edqm provides reference standards for the quality control of medicines and health care products in europe and worldwide. Find out how to subscribe, consult, and get help with the. Find and order reference standards for the european pharmacopoeia monographs from. The edqm also provides batch validity statements, safety data sheets and. Our new interactive 2025 catalogue is now available, offering instant access to sample data, scheme descriptions, application forms, and videos, plus a search function to quickly find what. Erythromycin c is a catalogue code for a reference standard used in european pharmacopoeia monographs. Edqm provides reference standards for the. The edqm provides chemical, herbal and biological reference standards for the tests and assays in the european pharmacopoeia. Learn how to participate in establishing and using the. If you select contains, all entries containing your search term will be returned. Access the full list here. We distribute chemical reference substances and reference standards from the world’s three leading pharmacopoeias: Eur.) offers a portfolio of more than 2 800 reference standards. Batch validity statements (bvss) for each reference standard;. Search for crs (certified reference substances) by name or code in the european pharmacopoeia catalogue. Access the full list here. Our new interactive 2025 catalogue is now available, offering instant access to sample data, scheme descriptions, application forms, and videos, plus. This document provides a list of european pharmacopoeia reference standards including the order, reference standard code, effective date, batch number, quantity per vial, sale unit,. Learn how to participate in establishing and using the. Access the full list here. Edqm provides reference standards for the quality control of medicines and health care products in europe and worldwide. Find out the. If you select contains, all entries containing your search term will be returned. Learn how to participate in establishing and using the. For example, if you enter. The united states pharmacopeia (usp), the european. This document provides a list of european pharmacopoeia reference standards including the order, reference standard code, effective date, batch number, quantity per vial, sale unit,. Download the pdf catalogue or use the online database with updated information and. The catalogue of the european pharmacopoeia (ph. Find and order reference standards for the european pharmacopoeia monographs from the edqm. Search by catalogue code, name, monograph or cas number to find the standard you need. This catalogue is updated on a daily. Search for crs (certified reference substances) by name or code in the european pharmacopoeia catalogue. Rs catalogue is updated on a daily basis and gives access not only to all the ph. We distribute chemical reference substances and reference standards from the world’s three leading pharmacopoeias: The edqm also provides batch validity statements, safety data sheets and. Find and buy online access to technical information for vehicle industry, such as spare part catalogues, workshop manuals, diagnostic programs and service manuals. Eur.) offers a portfolio of more than 2 800 reference standards.Ep technical catalogue
(PDF) EP Technical Catalogue for Fittings DOKUMEN.TIPS
What Is an EP? Why Should I Release One? Output
EP PDF CATALOGUE Instagram, Facebook Linktree
Rotork YTC YT1000R Electro Pneumatic Positioner Catalogue YTC INDIA
[EP] Catalog A4_R3 Erika Tevya Page 1 Flip PDF Online PubHTML5
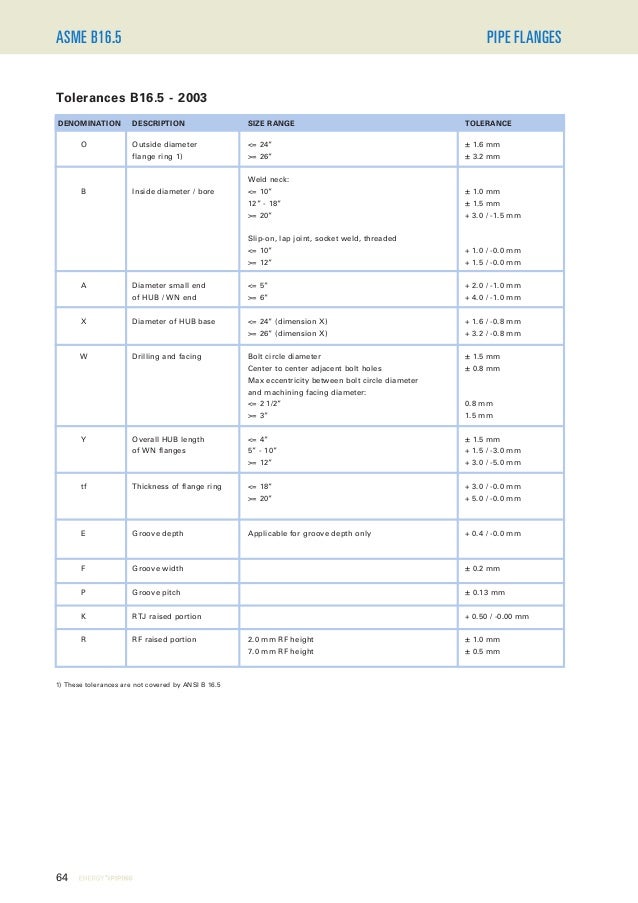
Ep technical catalogue
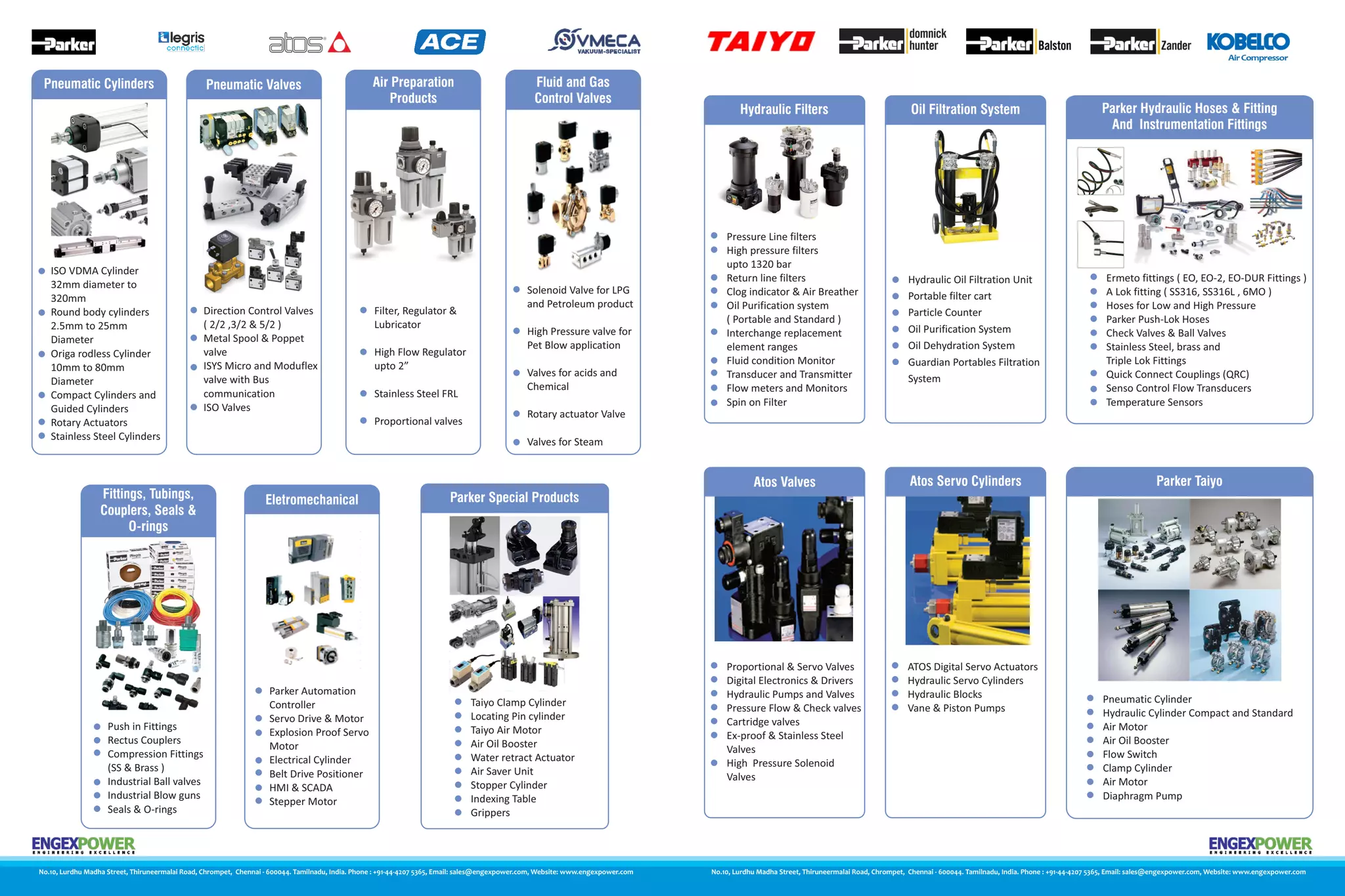
Ep catalogue PPT
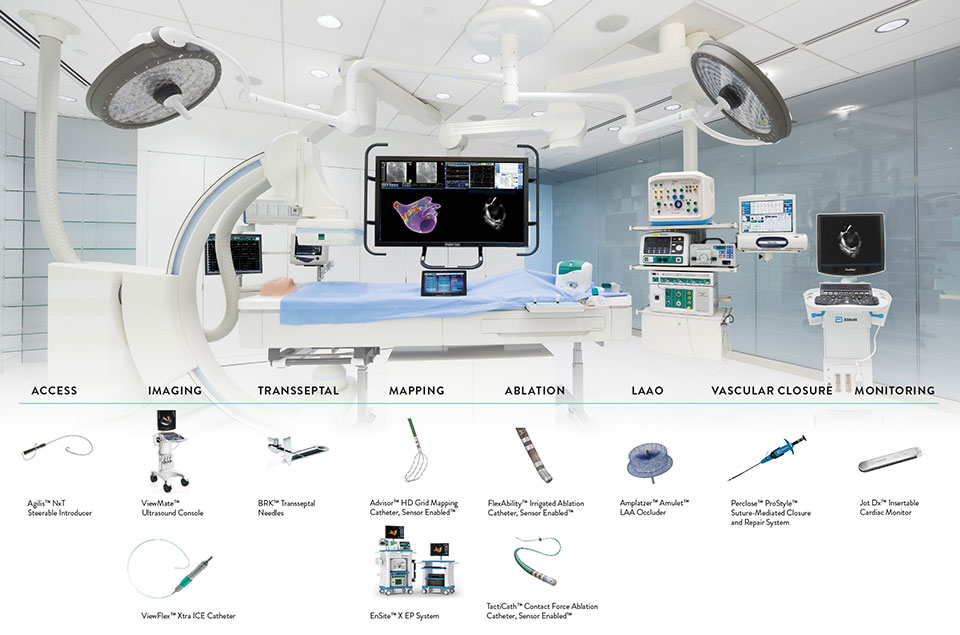
Abbott Ep Product Catalog Catalog Library
EP COLLECTION Cd made by me Contains The full catalogue of Ep's
Please Enter A Search Term And Select A Search Method Using The Drop Menus Below.
Find Information On Batch Number, Unit Quantity, Price, Shipping Conditions And.
Edqm Provides Reference Standards For The Quality Control Of Medicines And Health Care Products In Europe And Worldwide.
Our New Interactive 2025 Catalogue Is Now Available, Offering Instant Access To Sample Data, Scheme Descriptions, Application Forms, And Videos, Plus A Search Function To Quickly Find What.
Related Post:





![[EP] Catalog A4_R3 Erika Tevya Page 1 Flip PDF Online PubHTML5](https://online.pubhtml5.com/crlu/qsgu/files/large/1.jpg)